Ginsenoside Rg3 promotes regression from hepatic fibrosis through reducing inflammation-mediated autophagy signaling pathwayg
Xiangxiang Liu1, Xiaojie Mi1, Zi Wang1,2, Ming Zhang3, Jingang Hou1, Shuang Jiang1,2, Yingping Wang1,2, Chen Chen 4 and Wei Li1,2
Abstract
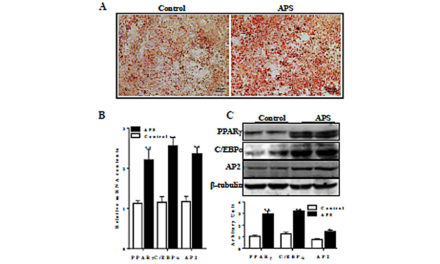
Inflammation and autophagy occur during hepatic fibrosis development caused by various pathogens, and effectively curbing of autophage may delay the occurrence of hepatic fibrosis. The current study aimed to unravel the inhibitory effects of Ginsenoside Rg3 (G-Rg3) on inflammation-mediated hepatic autophagy to curb hepatic fibrosis caused by thioacetamide (TAA)-induced subacute and chronic hepatic injury. TAA is mainly metabolized in the liver to cause liver dysfunction. After intraperitoneal injection of TAA for 4 or 10 weeks (TAA-chronic mouse models), severe inflammatory infiltration and fibrosis occurred in the liver. Treatment with G-Rg3 alleviated hepatic pathological changes and reversed hepatic fibrosis in the TAA-chronic models with decreased deposition of collagen fibers, reduced expression of HSCs activation marker (α-SMA), and reduced secretion of profibrogenic factors (TGF-β1). G-Rg3 decreased expressions of autophagy-related proteins in mice of TAA-chronic models. Notably, G-Rg3 inhibited the survival ofactivated rat hepatic stellate cells (HSC-T6), but had no cytotoxicity on human hepatocytes (L02 cell lines). G-Rg3 dose-dependently inhibited autophagy in vitro with less expression of p62 and fewer LC3a transformation into LC3b in inflammatory inducer lipopolysaccharide (LPS)-induced rat HSC-T6 cells. Furthermore, G-Rg3 enhanced the phosphorylation of phosphatidylinositol 3-kinase (PI3K) and protein kinase B (Akt) in vivo and in vitro. Besides, mTOR inhibitor Rapamycin and PI3K inhibitors LY294002 were employed in LPS-treated HSC-T6 cell cultures to verify that Rg3 partially reversed the increase in autophagy in hepatic fibrosis in vitro. Taken together, G-Rg3 exerted anti-fibrosis effect through the inhibition of autophagy in TAA-treated mice and LPS-stimulated HSC-T6 cells. These data collectively unravel that G-Rg3 may serve a promising anti-hepatic fibrosis drug.
(…)